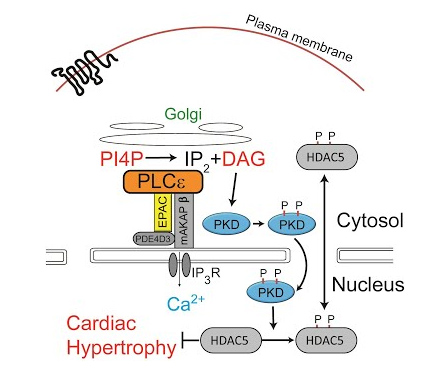
Cardiovascular
G protein coupled receptors play a very important role in the cardiovascular system where they regulate blood pressure and cardiac output in response to hormones such as adrenaline and angiotensin. During acute physiological responses these hormones are essential for stress and exercise responses, however; chronic stimulation of these pathways under conditions of cardiac damage due to myocardial infarction (MI) or high blood pressure leads to enlargement of the heart and development of heart failure.
We are investigating the molecular pathways in cardiac cells driven by chronic GPCR activation. There are changes in cardiac size and gene expression that can be seen both in whole animals and in isolated cardiac cells. We use genetic knockout animal models, live cell cardiomyocyte microscopic fluorescence imaging, in vitro biochemistry, and cell physiology to understand the detailed mechanisms that underlie GPCR driven cardiac hypertrophy. As a result of some of these studies we have developed novel strategies for treatment of heart failure. In collaboration with other laboratories in the CVC here at Michigan, and elsewhere, we are also studying other roles for GPCR signaling in cardiovascular function.
Representative relevant publications:
Golgi Localized β1-adrenergic Receptors Stimulate Golgi PI4P Hydrolysis by PLCε to Regulate Cardiac Hypertrophy
Nash CA, Wei W, Irannejad, R, Smrcka AV.
eLife. 2019 Aug 21
Compartmentalized cyclic nucleotides have opposing effects on regulation of hypertrophic phospholipase Cε signaling in cardiac myocytes.
Nash CA, Brown LM, Malik S, Cheng X, Smrcka AV.
J Mol Cell Cardiol. 2018 Jun 7;121:51-59
Phospholipase Cε Modulates Rap1 Activity and the Endothelial Barrier.
DiStefano PV, Smrcka AV, Glading AJ.
PLoS One. 2016 Sep 9;11(9):e0162338
G protein βγ subunits regulate cardiomyocyte hypertrophy through a perinuclear Golgi phosphatidylinositol 4-phosphate hydrolysis pathway.
Malik S, deRubio RG, Trembley M, Irannejad R, Wedegaertner PB, Smrcka AV.
Mol Biol Cell. 2015 Mar 15;26(6):1188-98
Regulation of phosphatidylinositol-specific phospholipase C at the nuclear envelope in cardiac myocytes.
Smrcka AV.
J Cardiovasc Pharmacol. 2015 Mar;65(3):203-10 (REVIEW)
Simultaneous adrenal and cardiac g-protein-coupled receptor-gβγ inhibition halts heart failure progression.
Kamal FA, Mickelsen DM, Wegman KM, Travers JG, Moalem J, Hammes SR, Smrcka AV, Blaxall BC.
J Am Coll Cardiol. 2014 Jun 17;63(23):2549-57
Phospholipase Cε hydrolyzes perinuclear phosphatidylinositol 4-phosphate to regulate cardiac hypertrophy.
Zhang L, Malik S, Pang J, Wang H, Park KM, Yule DI, Blaxall BC, Smrcka AV.
Cell. 2013 Mar 28;153(1):216-27
Phospholipase C epsilon scaffolds to muscle-specific A kinase anchoring protein (mAKAPbeta) and integrates multiple hypertrophic stimuli in cardiac myocytes.
Zhang L, Malik S, Kelley GG, Kapiloff MS, Smrcka AV.
J Biol Chem. 2011 Jul 1;286(26):23012-21
Epac and phospholipase Cepsilon regulate Ca2+ release in the heart by activation of protein kinase Cepsilon and calcium-calmodulin kinase II.
Oestreich EA, Malik S, Goonasekera SA, Blaxall BC, Kelley GG, Dirksen RT, Smrcka AV.
J Biol Chem. 2009 Jan 16;284(3):1514-22
View all publications here.
G protein signaling in the central nervous system
G protein coupled receptors are highly expressed in the central nervous system and regulate neurotransmission and development. We are actively investigating these functions in two projects. 1) Development of small molecule drug like molecules that bind to G proteins to modify the actions of opioid receptors. The goal of this project in collaboration with the Jutkiewicz Lab is to improve the safety of opioids as critical pain medications. 2) Understanding the roles of G protein mutations that underly developmental epileptic encephalopathies to develop new therapeutic strategies. This project has been pushed forward by the identification of novel G protein signaling partners using a novel proteomics based method.
Representative relevant publications:
Biasing Gβγ Downstream Signaling with Gallein Inhibits Development of Morphine Tolerance and Potentiates Morphine-Induced nociception in a Tolerant State. Molecular Pharmacology, May 2024
Coincident Regulation of PLC β Signaling by Gq-Coupled and μ-Opioid Receptors Opposes Opioid-Mediated Antinociception. Molecular Pharmacology, December 2022.
A network of Gαi signaling partners is revealed by proximity labeling proteomics analysis and includes PDZ-RhoGEF. Science Signaling, January 2022
View all publications here.
Drug Discovery
30-40% of the drugs on the market target GPCRs. This is largely because GPCRs have the capacity to bind drugs and regulate a wide range of cellular physiologies. Our laboratory has promoted and developed the idea that targeting the G protein after the GPCR could be a novel and more efficacious strategy for the treatment of various conditions including heart failure, inflammation, pain and cancer. We use high-throughput chemical screening to identify “drug like” inhibitors of G protein βγ subunit signaling. Compounds identified so far have been highly effective in mouse models of pain, inflammation and heart failure. In collaboration with the Center for Chemical Genomics here at Michigan we are performing larger scale high throughput screens for new and better Gβγ inhibitors that will be optimized and tested with the goal of developing novel therapeutic agents for improving the therapeutic efficacy of opioids while avoiding detrimental side effects. This process involves chemistry, biochemistry, cell biology and whole animal studies.
Representative relevant publications:
Inhibition of G Protein βγ Subunit Signaling Abrogates Nephritis in Lupus-Prone Mice.
Rangel-Moreno J, To JY, Owen T, Goldman BI, Smrcka AV, Anolik JH.
Arthritis Rheumatol. 2016 Sep;68(9):2244-56
A chemical biology approach demonstrates G protein βγ subunits are sufficient to mediate directional neutrophil chemotaxis.
Surve CR, Lehmann D, Smrcka AV. J Biol Chem. 2014 Jun 20;289(25):17791-801.
Molecular targeting of Gα and Gβγ subunits: a potential approach for cancer therapeutics.
Smrcka AV. Trends Pharmacol Sci. 2013 May;34(5):290-8 REVIEW
Taking the heart failure battle inside the cell: small molecule targeting of Gβγ subunits.
Kamal FA, Smrcka AV, Blaxall BC. J Mol Cell Cardiol. 2011 Oct;51(4):462-7 REVIEW
Small molecule disruption of G beta gamma signaling inhibits the progression of heart failure.
Casey LM, Pistner AR, Belmonte SL, Migdalovich D, Stolpnik O, Nwakanma FE, Vorobiof G, Dunaevsky O, Matavel A, Lopes CM, Smrcka AV, Blaxall BC. Circ Res. 2010 Aug 20;107(4):532-9
Small molecule disruption of G protein beta gamma subunit signaling inhibits neutrophil chemotaxis and inflammation.
Lehmann DM, Seneviratne AM, Smrcka AV. Mol Pharmacol. 2008 Feb;73(2):410-8.
Differential targeting of Gbetagamma-subunit signaling with small molecules.
Bonacci TM, Mathews JL, Yuan C, Lehmann DM, Malik S, Wu D, Font JL, Bidlack JM, Smrcka AV. Science. 2006 Apr 21;312(5772):443-6
View all publications here.
Phospholipase C signaling
One of the major signaling pathways regulated by cell surface receptors in virtually all cells and tissues is hydrolysis of the membrane phospholipid phosphatidylinositol 4,5 bisphosphate (PIP2) by Phospholipase C (PLC). This reaction controls calcium signals and protein kinase activation which in turn have profound effects on cellular physiology. Our laboratory has been interested in the biochemistry and physiology of this signaling pathway for many years. Some examples of projects include
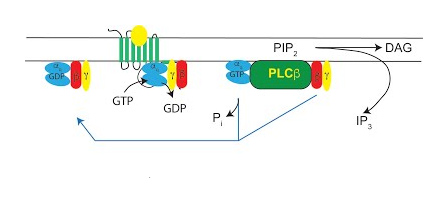
- PLC in cardiac hypertrophy. Using novel genetic mouse knockout models generated in our laboratory we have shown that deletion of PLC signaling in cardiac myocytes protects mice against stress-induced heart failure. We are using new mouse models and AAV vector systems to further dissect these processes in vivo.
- Cardiomyocyte PLC signaling networks. Using isolated cardiomyocytes we use biochemistry and live cell imaging to map PLC signaling networks that regulate cardiac contraction and growth.
- PLC signaling in other biologies. With our PLC knockout mice and Crispr cell lines we are in position to examine multiple roles in physiology and disease.
- PLC biochemistry. We use molecular biology, cell biology and in vitro protein purification to understand the mechanisms that regulate PLC enzymatic activity.
Representative relevant publications:
Activation of Phospholipase C β by Gβγ and Gαq Involves C-Terminal Rearrangement to Release Autoinhibition
Fisher IJ, Jenkins ML, Tall GG, Burke JE, Smrcka AV.
Structure, 2020 May 4
Golgi Localized β1-adrenergic Receptors Stimulate Golgi PI4P Hydrolysis by PLCε to Regulate Cardiac Hypertrophy
Nash CA, Wei W, Irannejad, R, Smrcka AV.
eLife. 2019 Aug 21
Phosphatidylinositol 4-phosphate is a major source of GPCR-stimulated phosphoinositide production.
de Rubio RG, Ransom RF, Malik S, Yule DI, Anantharam A, Smrcka AV.
Sci Signal. 2018 Sep 11;11(547).
G protein βγ subunits directly interact with and activate phospholipase Cϵ.
Madukwe JC, Garland-Kuntz EE, Lyon AM, Smrcka AV.
J Biol Chem. 2018 Apr 27;293(17):6387-6397
Phospholipase Cε Modulates Rap1 Activity and the Endothelial Barrier.
DiStefano PV, Smrcka AV, Glading AJ.
PLoS One. 2016 Sep 9;11(9):e0162338
Phospholipase C-ε signaling mediates endothelial cell inflammation and barrier disruption in acute lung injury.
Bijli KM, Fazal F, Slavin SA, Leonard A, Grose V, Alexander WB, Smrcka AV, Rahman A.
Am J Physiol Lung Cell Mol Physiol. 2016 Aug 1;311(2):L517-24.
Thrombin promotes sustained signaling and inflammatory gene expression through the CDC25 and Ras-associating domains of phospholipase Cϵ.
Dusaban SS, Kunkel MT, Smrcka AV, Brown JH.
J Biol Chem. 2015 Oct 30;290(44):26776-83
M3 muscarinic receptor interaction with phospholipase C β3 determines its signaling efficiency.
Kan W, Adjobo-Hermans M, Burroughs M, Faibis G, Malik S, Tall GG, Smrcka AV.
J Biol Chem. 2014 Apr 18;289(16):11206-18
Phospholipase Cε hydrolyzes perinuclear phosphatidylinositol 4-phosphate to regulate cardiac hypertrophy.
Zhang L, Malik S, Pang J, Wang H, Park KM, Yule DI, Blaxall BC, Smrcka AV.
Cell. 2013 Mar 28;153(1):216-27
Phospholipase C epsilon links G protein-coupled receptor activation to inflammatory astrocytic responses.
Dusaban SS, Purcell NH, Rockenstein E, Masliah E, Cho MK, Smrcka AV, Brown JH.
Proc Natl Acad Sci U S A. 2013 Feb 26;110(9):3609-14
Epac2-dependent mobilization of intracellular Ca²+ by glucagon-like peptide-1 receptor agonist exendin-4 is disrupted in β-cells of phospholipase C-ε knockout mice.
Dzhura I, Chepurny OG, Kelley GG, Leech CA, Roe MW, Dzhura E, Afshari P, Malik S, Rindler MJ, Xu X, Lu Y, Smrcka AV, Holz GG.
J Physiol. 2010 Dec 15;588(Pt 24):4871-89.
Epac and phospholipase Cepsilon regulate Ca2+ release in the heart by activation of protein kinase Cepsilon and calcium-calmodulin kinase II.
Oestreich EA, Malik S, Goonasekera SA, Blaxall BC, Kelley GG, Dirksen RT, Smrcka AV.
J Biol Chem. 2009 Jan 16;284(3):1514-22
View all publications here.